What is Photochemistry and how is involved with the development of film?
Photochemistry is the process by which electromagnetic energy causes chemical changes to matter. It is involved with the development of film because the chemical reactions allow the images developed from the film to obtain the colours it has.
Each roll of film is comprised of 4 components
Silver-halide Crystals & Spectral sensitizers (Heart of the Photographic film)
Silver-halide crystals
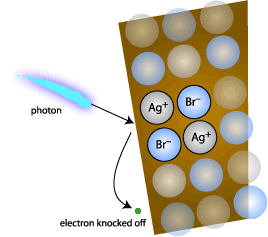
The Structure (How it forms) : Crystal lattice of a silver bromide crystal -> contains ions of Bromine (Br-) & Silver (Ag+)
Photochemistry is the process by which electromagnetic energy causes chemical changes to matter. It is involved with the development of film because the chemical reactions allow the images developed from the film to obtain the colours it has.
Each roll of film is comprised of 4 components
- The Base: is the component that all the other layers and chemicals within the roll of film is clung on to. The base is essential to the film because without it, the film’s structure would be very frail and more prone to tear apart due to the mechanical strain occurring inside the camera.
- The Adhesive: is the component that binds all the individual photochemical layers together which in result binds these layers to the base material of the film.
- The Emulsion: Is a light sensitive colloid. It is known as a colloid because microscopically dispersed insoluble particles are suspended throughout this layer. This layer consists of sub-micron sized grains of silver halide crystals dispersed in a fluid (gelatin). This component of the film is vital because it holds key to the photochemical processes occuring inside the film.
- The Supercoat: is a protective coating that keeps the film from being damaged during the development of the film.
Silver-halide Crystals & Spectral sensitizers (Heart of the Photographic film)
Silver-halide crystals
- Manufactured by: combining silver-nitrate and halide salts (chloride, bromide and iodide) in very intricate ways that result in the formation of different sizes of crystals (grain-like)
- are chemically modified on their surface to increase their light sensitivity
- are only sensitive to the blue portion of the spectrum which is not efficient in camera film because it only produces mere portions of the image
The Structure (How it forms) : Crystal lattice of a silver bromide crystal -> contains ions of Bromine (Br-) & Silver (Ag+)
- Film's exposure to light: extra electron from bromine ion is released
- Free electron attaches to positively charged silver ion
- Silver ion + free electron -> Metallic Silver
- This reaction occurs all over the layers and planes of the emulsion which creates a faded latent image on the film
Spectral sensitizers
- added to the surface of silver-halide crystal because it gives them to ability to be more sensitive to other colours of the visible spectrum (blue, green & red light)
- Transforms faded latent image on film made by silver-halide crystal to a visible image
The Photochemical Process
Before (When capturing the image):
A short exposure image formed by the lens of the film camera produces a slight chemical change which is proportionate to the amount of light absorbed by each grain of silver crystal halide at different intensities in the emulsion of the film. An invisible latent image in the emulsion is then formed which can be chemically developed into a visible photograph.
During (The chemical reaction):
The photochemical process involves turning the invisible latent image into a visible latent image on the film, this is completed through the chemical reaction of applying spectral sensitizers to the surface of silver-halide crystals.
- Spectral sensitzer molecules attach to the silver-halide crystal
- Transfer the energy from a red, green, or blue photon to the silver-halide crystal as a photo-electron (photon) & silver atom is knocked of of crystal lattice
After (The development) :
- The film is then development in a dark room which is a room that is almost completely dark but it is illuminated with an amber light-brown light known as a safelight
- The darkroom allows the processes of light sensitive materials such as film, the safelight is very suitable for darkrooms and the development of film because it provides illumination only from certain parts of the visible spectrum to which the photographic image being produced is almost completely insensitive towards .
- After the chemical process, spectral sensitization has been increased so when the film is developed, colour will appear.
- When photographs are developed, they can appear more red than blue or green or vice versa because a region of silver atoms may have held a higher concentration of that certain colour during the chemical reaction